Algae is a diverse group of mostly-aquatic eukaryotic organisms (their cells have a nucleus which contains genetic material). They contain chlorophyll (pigments that absorb light) and photosynthesize (they produce oxygen), but are not plants. They can be unicellular (composed of just one cell) or multicellular (composed of multiple cells). They dominate the flora of marine and freshwater ecosystems. It is probable that there is less diversity of algae in the Arctic than in warmer waters.
Brown algae (class Phaeophyceae) are a group of almost exclusively marine algae. This group contains many seaweeds (large marine algae) of temperate and Arctic seas and oceans. They dominate Arctic fjord environments at shallow depths. These seaweeds can be very large, such as giant kelp which form underwater forests. Brown algae have blades (similar to leaves) that grow on stipes (equivalent to stems) and they are attached to the sea floor by a holdfast. Some have bladders (small pockets of air) on their blades which help to make the blades float towards the surface to absorb more light. Examples of North Atlantic Arctic brown algae include:
- Oarweed Laminaria digitata. Like other kelp, oarweed looks like long brown ribbons swaying in the water. Five to eight blades can grow on one stipe, and the seaweed can grow up to two meters. Orweed is found in the North Atlantic from the Canary Islands to Svalbard, including Greenland, Iceland, the Faroe Islands and Fennoscandia. It is cultivated in Ireland and France and can be used in food production, cosmetics, pharmaceuticals, and as feed for edible marine invertebrates such as abalone and sea urchins.
- Rockweed Ascophyllum nodosum. This large cold water brown algae can grow up to two meters and has egg-shaped air bladders. It is present from southern Greenland to Portugal, including Iceland, the Faroe Islands, and Fennoscandia. Like oarweed, it can be used as animal feed or to extract algnic acid which is used in multiple industries. It often grows at the same depth as Bladder wrack Fucus vesiculosus.
- Bladder wrack Fucus vesiculosus. This large brown algae can be recognized from the pair of bladders growing along the blades. It is found in Greenland, Svalbard (in a dwarf form), Iceland, the Faroe Islands, and Fennoscandia.
Red algae (phylum Rhodophyta) are mostly marine, very diverse, and can grow deep as they don't need as much light as brown or green algae. Some red algae are coralline, meaning that they develop calcium branches that look like coral (which is formed by very small invertebrate animals).
- Dulse Palmaria palmata. Dulse is a dark red red algae growing in thin blades with lengths up to 50 centimeters. It is found on both sides of the North Atlantic, and from Portugal to Svalbard. It is also present in Iceland, the Faroe Islands, and Fennoscandia. It has been used a food for centuries, especially in Ireland, and it has also been consumed in Iceland, Norway, and Scotland. It is still cultivated and used as food in various countries.
- Irish moss Chondrus crispus, also called Carrageen. Irish moss is yellow-green to red and has thick, flat, branching blades. It is found on both sides of the Atlantic, from Iceland to Portugal including the Faroe Islands. This red algae is also used in the food industry for extracting carrageenan, which has gelling properties.
- Maerl such as Lithothamnion glaciale. Maerl is a group of coralline red algae that is common in the North Atlantic from Portugal to Svalbard. Maerl forms an important habitat for marine animals and algae on the sea floor, but it has been exploited in places such as Ireland and France to use as fertilizer. Dredging and harvesting damages and destroys maerl faster than it can grow. Lithothamnion glaciale is a species of maerl which is found in the northern Pacific and the northern Atlantic including Canada, Iceland, the Faroe Islands, Greenland, Svalbard, and the British Isles. It is considered the most abundant maerl species in the North Atlantic.
Green algae (class Chlorophyta) are the closest relatives to the land plants among algae groups, as it is appears land plants may have evolved from a type of green algae. Like land plants, they contain both chlorophyll a and chlorophyll b, which gives them a bright green color. Many species of green algae are only found in freshwater, but other species are adapted to marine life. Some green algae are microscopic, and because of climate change, some green micro-algae are becoming prone to algal blooms (rapid increase in the population of algae) on the Arctic ice sheet, for example in Greenland. This makes the ice darker, in turn increasing sun absorption and thus making the glaciers melt faster.
- Green tarantula weed Acrosiphonia arcta. This green algae forms thin filaments that grow up to 10 centimeters. These filaments can be seen when the algae is submerged, but in dry conditions they cannot be distinguished and the algae is shapeless. It has a circumpolar distribution, and is also found in the Southern Hemisphere down to the Antarctic. In the North Atlantic, it is found in Svalbard, Scandinavia, Greenland, and Iceland, where it is one of the most common green algae species.
- Glaucous brick-weed Chaetomorpha melagonium. Glaucous brick-weed grows in separated hair-like strands made up of one single row of cells. It is found in the North Atlantic including in Svalbard, Scandinavia, the Faroe Islands, and Iceland.
- Sea lettuce Ulva prolifera. This species is found almost worldwide, including Svalbard, Greenland, Iceland, Scandinavia, and the Faroe Islands. In some areas, such as the Yellow sea, it is an non-native species which has been causing green-tides (when seaweeds grow extremely fast in a specific area). These green-tides can have disastrous consequences for local ecology, causing decreasing levels of oxygen in the water as well as water acidification.
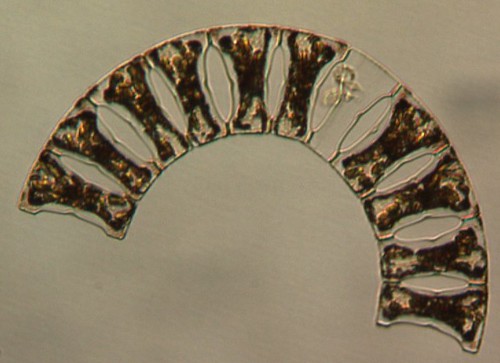
Diatoms are unicellular microalgae that can grow up to 2 millimeters long. They are protected by thin shells made of silica (a hard crystal material which is also a major component in sand). Diatoms are the main primary producers in the Arctic Ocean, which means that they are the organisms that collectively produce the most organic matter in this region by using the sun's energy. Through this process, photosynthesis, they also release 20-30% of the oxygen we breathe. In the ocean, diatoms are part of phytoplankton, meaning they are free-floating in the water. They are the base of the food chain, being eaten by zooplankton (marine microanimals that are free-floating in the water) which are in turn eaten by fish and other marine animals. Diatoms are extremely well adapted to the cold, nutrient rich, and stormy waters of the poles, and they are even common inside sea ice. Examples of North Atlantic Arctic diatoms include:
- Fragilariopsis oceanica. This Arctic species has a circumpolar distribution, being found in sea water and sea ice in Arctic, Pacific, and Atlantic oceans. This includes around Iceland, Svalbard, and Greenland, where it can be the most abundant diatom species in some areas.
- Nitzschia frigida. This species is found in temperate to Arctic waters in temperate and Arctic waters of the North Atlantic. In the Arctic, it can be present both in water and ice.
- Eucampia groenlandica. This water and ice Arctic species is distributed in the North Atlantic from northern France and the northeastern coast of the U.S. to High Arctic sea ice. It is most frequent in the waters between Greenland and Canada.
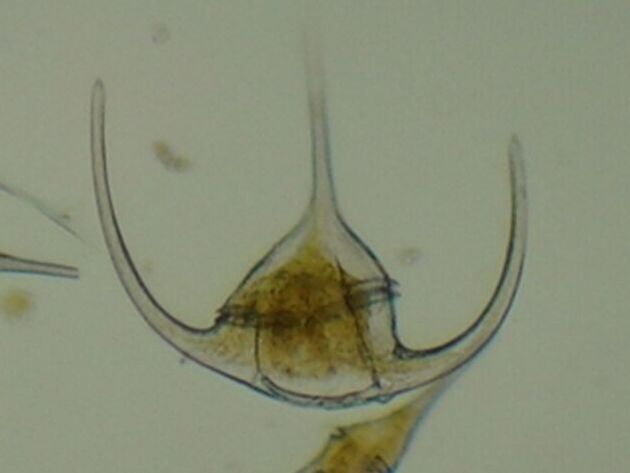
Dinoflagellates and diatoms are the two main classes that form the phytoplankton (free floating microalgae in the ocean or freshwater). Dinoflagellates are very interesting, because they are unicellular but complex. They have complex shells, and can swim in the water on a microscopic level thanks to their flagella (small tentacle-like organs that are used for movement). Some species are only autotrophic (they produce their own energy through photosynthesis), some are heterotrophic (they consume other organisms for energy), some are mixotrophic (they gain energy through photosynthesis as well as through consuming other organisms), and some are parasitic, specifically in fish. Unlike diatoms, dinoflagellates are not very diverse in boreal and Arctic areas, with no known species endemic to the Arctic. Arctic phytoplankton is mostly composed of diatoms, but in some areas such as the Norwegian sea, dinoflagellates can outnumber diatoms in the summer. Some examples of Arctic dinoflagellates include:
- Ceratium arcticum. Ceratium arcticum has a circumpolar distribution and is found from temperate to Arctic waters of the North Atlantic. It is found around Iceland, Scandinavia including the Baltic sea, the Faroe Islands, and Greenland. In the northwestern Atlantic, its abundance has changed drastically since the 1960's, with increased blooms in the winter and decreased abundance in the autumn.
- Dinophysis norvegica. This species is found almost exclusively in the North Atlantic, but is also present between Alaska and eastern Siberia. In the East Atlantic Arctic, it is found around Iceland, Scandinavia including the Baltic sea, the Faroe Islands, Greenland, and the southern coast of Svalbard. This dinoflagellate is mixotrophic, feeding on other microalgae as a primary source of energy and using photosynthesis as a secondary source. Because shellfish feed on them, some species of the genus Dinophysis (such as Dinophysis acuta and possibly Dinophysis norvegica) can cause shelfish to become toxic to humans and animals. In Norway, they are responsible for causing diarrhea in people eating mussels, especially in the autumn.
- Alexandrium ostenfeldii. Alexandrium ostenfeldii is bioluminescent dinoflagellate with a circumpolar distribution, and it is found in the East Atlantic Arctic around Scandinavia and the Faroe Islands. Many species in the Alexandrium genus cause can cause paralytic shellfish poisoning in humans and animals after algal blooms. Due to climate change, Alexandrium ostenfeldii has been causing more and more severe and frequent toxic algal blooms, also called red tides, in the northern Baltic Sea.
Pictures
- "Ascophyllum nodosum" by wanderflechten is licensed under CC BY-NC-ND 2.0.
- "Laminaria digitata" by cquintin is licensed under CC BY-NC 2.0.
- "Ascophyllum nodosum" by wanderflechten is licensed under CC BY-NC-ND 2.0.
- "Fucus vesiculosus" by Hans Hillewaert is licensed under CC BY-NC-ND 2.0.
- "Dulse" by Charles.Andre is licensed under CC BY-NC-SA 2.0.
- "Chondrus crispus" by cquintin is licensed under CC BY-NC 2.0.
- "Thick coralline crust (mainly Lithothamnion glaciale) with anemones (Metridium senile), Newfoundland" by Derek Keats is licensed under CC BY 2.0.
- "Acrosiphonia arcta 2 Crouan.jpg" by Pierre-Louis Crouan & Hippolyte-Marie Crouan is in the public domain.
- "Chaetomorpha melagonium (F.Weber & D.Mohr) Kützing" by Colin Bates is copyrighted by Coastal Imageworks.
- "File:Meersalat-Ulva-lactuca.jpg" by H. Krisp is licensed under CC BY 3.0.
- "Fragilariopsis kerguelensis" by ZEISS Microscopy is licensed under CC BY-NC-ND 2.0.
- "NITZSCHIA SIGMOIDEA: ¡CUIDADO, QUE VIENE EL COCO!" by PROYECTO AGUA** /** WATER PROJECT is licensed under CC BY-NC-SA 2.0.
- "fish1890" by NOAA Photo Library is licensed under CC BY 2.0.
- "Phytoplankton - Ceratium species - Mississippi Department of Marine Resources ~ Shellfish Bureau" by Mississippi Department of Marine Resources is licensed under CC BY-NC-ND 2.0.
- "File:Dinophysis norvegica.jpg" by Fisheries and Oceans Canada, Dalhousie University, Rajashree Gouda is licensed under CC BY-SA 4.0.
- "Alexandrium bloom Naval Weapons Station" by VA Institute of Marine Science (VIMS) is licensed under CC BY-NC-ND 2.0.
References
- Boedeker, C., Leliaert, F., & Zuccarello, G. C. (2017). Genetic diversity and biogeography in Chaetomorpha melagonium (Ulvophyceae, Cladophorales) based on internal transcribed spacer (ITS rDNA) sequences. Botanica Marina, 60(3), 319–325. https://doi.org/10.1515/BOT-2016-0129
- Büdenbender, J., Riebesell, U., & Form, A. (2011). Calcification of the Arctic coralline red algae Lithothamnion glaciale in response to elevated CO 2. Marine Ecology Progress Series, 441, 79–87. https://doi.org/10.3354/MEPS09405
- Carty, S., Parrow, M.W. (2015). Dinoflagellates. In Freshwater Algae of North America (Second Edition).
- Carvalho, W. F., Minnhagen, S., & Granéli, E. (2008). Dinophysis norvegica (Dinophyceae), more a predator than a producer? Harmful Algae, 7(2), 174–183. https://doi.org/10.1016/J.HAL.2007.07.002
- Daniëls, F.J.A., Gillespie, L.J., Poulin, M., Afonina, O.M., Alsos, I.G., Aronsson, M., Bültmann, H., Ickert-Bond, S., Konstantinova, N.A., Lovejoy, C., Väre, H., Bakke Westergaard, K. (2013). Flora. In Arctic Biodiversity Assessment (pp. 310–352). https://www.caff.is/assessment-series/arctic-biodiversity-assessment/214-arctic-biodiversity-assessment-2013-chapter-9-flora
- Edwards, M., & Watson, L. (2011). Cultivating Laminaria digitata. Irish Sea Fisheries Board - Aquaculture Explained, 26. https://www.researchgate.net/publication/281611094_Cultivating_Laminaria_digitata
- Fredriksen, S., Gabrielsen, T. M., Kile, M. R., & Sivertsen, K. (2015). Benthic algal vegetation in Isfjorden, Svalbard. Polar Research, 34(1). https://doi.org/10.3402/POLAR.V34.25994/SUPPL_FILE/ZPOR_A_11818905_SM0001.PDF
- Fredriksen, S., Kile, M.R. (2012). The algal vegetation in the outer part of Isfjorden, Spitsbergen: revisiting Per Svendsen's sites 50 years later. Polar Research 31, 17538, DOI: 10.3402/polar.v31i0.17538. https://polarresearch.net/index.php/polar/article/view/2602/html.
- Grall, J., Hall-Spencer, J.M. (2003). "Problems facing maerl conservation in Brittany" (PDF). Journal of Aquatic Conservation: Marine and Freshwater Ecosystems. 13: 55–64.
- Hall-Spencer, J., Kelly, J., & Maggs, C. A. (2010). Background document on maerl beds. https://www.researchgate.net/publication/230661931_Background_document_on_maerl_beds
- Hill, J.M. (2008). In Tyler-Walters H. and Hiscock K. Marine Life Information Network: Biology and Sensitivity Key Information Reviews, [on-line]. Plymouth: Marine Biological Association of the United Kingdom.
- Ascophyllum nodosum Knotted wrack. [accessed 24-08-2022]. Available from: https://www.marlin.ac.uk/species/detail/1336
- Palmaria palmata Dulse. [accessed 24-08-2022]. Available from: https://www.marlin.ac.uk/species/detail/1405
- IrishSeaweeds.com. "Kelp (Laminaria digitata)" Accessed 24 August 2022. https://irishseaweeds.com/kelp-laminaria-digitata/
- Jackson, A. 2003. Lithothamnion glaciale Maerl. In Tyler-Walters H. and Hiscock K. Marine Life Information Network: Biology and Sensitivity Key Information Reviews, [on-line]. Plymouth: Marine Biological Association of the United Kingdom. [cited 24-08-2022]. Available from: https://www.marlin.ac.uk/species/detail/1314
- Jiang, S., Seidenkrantz, M.S., Knudsen, K.L., Eiriksson, J. (2000). Diatom surface sediment assemblages around Iceland and their relationships to oceanic environmental variables. Marine Micropaleontology. 41(1-2), 73–96. 10.1016/S0377-8398(00)00053-0
- Johns, D. G., Edwards, M., Richardson, A., & Spicer, J. I. (2003). Increased blooms of a dinoflagellate in the NW Atlantic. Marine Ecology Progress Series, 265, 283–287. https://doi.org/10.3354/MEPS265283
- Kremp, A., Lindholm, T., Dreßler, N., Erler, K., Gerdts, G., Eirtovaara, S., & Leskinen, E. (2009). Bloom forming Alexandrium ostenfeldii (Dinophyceae) in shallow waters of the Åland Archipelago, Northern Baltic Sea. Harmful Algae, 8(2), 318–328. https://doi.org/10.1016/J.HAL.2008.07.004
- Lundholm, N., & Hasle, G. R. (2010). Fragilariopsis (Bacillariophyceae) of the Northern Hemisphere - Morphology, taxonomy, phylogeny and distribution, with a description of F. pacifica sp. nov. Phycologia, 49(5), 438–460. https://doi.org/10.2216/09-97.1
- Mock, T. (2017). A journey into the fascinating world of polar diatoms. Nature Portfolio Ecology & Evolution Community. https://ecoevocommunity.nature.com/posts/14083-the-genome-sequence-of-a-polar-diatom-explains-evolutionary-adaptation-to-one-of-the-most-extreme-and-threatened-ecosystem-on-earth
- Naustvoll, L. J., Gustad, E., & Dahl, E. (2012). Monitoring of Dinophysis species and diarrhetic shellfish toxins in Flødevigen Bay, Norway: inter-annual variability over a 25-year time-series. Https://Doi.Org/10.1080/19440049.2012.714908, 29(10), 1605–1615. https://doi.org/10.1080/19440049.2012.714908
- Nielsen, R., & Gunnarsson, K. (2001). Seaweeds of the Faroe Islands - An annotated checklist. Annales Societatis Scientiarum Færoensis, 49. https://timarit.is/page/931863#page/n39/mode/2up
- Okolodkov, Y. B., & Dodge, J. D. (1996). Biodiversity and biogeography of planktonic dinoflagellates in the Arctic Ocean. Journal of Experimental Marine Biology and Ecology, 202(1), 19–27. https://doi.org/10.1016/0022-0981(96)00028-7
- Peña, V., Bárbara, I., Grall, J., Maggs, C. A., & Hall-Spencer, J. M. (2014). The diversity of seaweeds on maerl in the NE Atlantic. Marine Biodiversity, 44(4), 533–551. https://doi.org/10.1007/S12526-014-0214-7
- Pereira, L. (2015). Seaweed flora of the european north atlantic and mediterranean. Springer Handbook of Marine Biotechnology, 65–178. https://doi.org/10.1007/978-3-642-53971-8_6
- Rayment, W.J. & Pizzola, P.F. 2008. Chondrus crispus Carrageen. In Tyler-Walters H. and Hiscock K. Marine Life Information Network: Biology and Sensitivity Key Information Reviews, [on-line]. Plymouth: Marine Biological Association of the United Kingdom. [cited 24-08-2022]. Available from: https://www.marlin.ac.uk/species/detail/1444
- Spaulding, S. A., Potapova, M. G., Bishop, I. W., Lee, S. S., Gasperak, T. S., Jovanoska, E., Furey, P. C., & Edlund, M. B. (2021). Diatoms.org: supporting taxonomists, connecting communities. Diatom Research, 36(4), 291–304. https://doi.org/10.1080/0269249X.2021.2006790
- Sun, Y., Liu, J., Xia, J., Tong, Y., Li, C., Zhao, S., Zhuang, M., Zhao, X., Zhang, J., & He, P. (2022). Research development on resource utilization of green tide algae from the Southern Yellow Sea. Energy Reports, 8, 295–303. https://doi.org/10.1016/J.EGYR.2022.01.168
- Søreide, J. E., Vader, A., Pitusi, V., Andreasen, M., Marquardt, M., Gradinger, R., Gerland, S., & Hop, H. (2021). Fjord ice in Svalbard — an important kindergarten for seafloor animal babies. The Independent Barents Observer. https://thebarentsobserver.com/en/node/8943
- Telnes, K. (2020). Green Tarantula Weed - Acrosiphonia arcta. The Marine Flora and Fauna of Norway. http://www.seawater.no/flora/green/arcta.html
- The Seaweed Site: information on marine algae.
- "Ascophyllum nodosum (L.) Le Jolis". [accessed 24-08-2022]. https://www.seaweed.ie/descriptions/Ascophyllum_nodosum.php
- "Chondrus crispus Stackhouse". [accessed 24-08-2022]. https://www.seaweed.ie/descriptions/Chondrus_crispus.php
- "Laminaria digitata (Hudson) J.V. Lamouroux". [accessed 24-08-2022]. https://www.seaweed.ie/descriptions/Laminaria_digitata.php
- "Palmaria palmata (Linnaeus) Weber & Mohr". [accessed 24-08-2022]. https://www.seaweed.ie/descriptions/Palmaria_palmata.php
- Thyrring, J., Wegeberg, S., Blicher, M. E., Krause-Jensen, D., Høgslund, S., Olesen, B., Jozef, W., Mouritsen, K. N., Peck, L. S., & Sejr, M. K. (2021). Latitudinal patterns in intertidal ecosystem structure in West Greenland suggest resilience to climate change. Ecography, 44(8), 1156–1168. https://doi.org/10.1111/ECOG.05381
- Valtýsson, H. Þ., Bogason, E., & dos Santos, E. (n.d.). Algae. Arctic Portal - Sea Iceland. Retrieved August 23, 2022, from https://seaiceland.is/about-us
- Valtýsson, H. Þ., Bogason, E. Green Tarantula Weed. Sjávarlíf – Life in the Icelandic Waters. Retrieved August 23, 2022, from https://www.sjavarlif.is/en/project/green-tarantula-weed/
- von Quillfeldt, C. H. (2000). Common Diatom Species in Arctic Spring Blooms: Their Distribution and Abundance. Botanica Marina, 43(6), 499–516. https://sci-hub.se/10.1515/BOT.2000.050
- White, N. 2008. Fucus vesiculosus Bladder wrack. In Tyler-Walters H. and Hiscock K. Marine Life Information Network: Biology and Sensitivity Key Information Reviews, [on-line]. Plymouth: Marine Biological Association of the United Kingdom. [cited 24-08-2022]. Available from: https://www.marlin.ac.uk/species/detail/1330
- Williamson, C. J., Anesio, A. M., Cook, J., Tedstone, A., Poniecka, E., Holland, A., Fagan, D., Tranter, M., & Yallop, M. L. (2018). Ice algal bloom development on the surface of the Greenland Ice Sheet. FEMS Microbiology Ecology, 94(3), 25. https://doi.org/10.1093/FEMSEC/FIY025
- WoRMS - World Register of Marine Species.
- "Eucampia groenlandica Cleve, 1896". [accessed 25-08-2022]. https://www.marinespecies.org/aphia.php?p=taxdetails&id=157430#distributions
- "Nitzschia frigida Grunow, 1880". [accessed 25-08-2022]. https://www.marinespecies.org/aphia.php?p=taxdetails&id=175400#distributions
- Zhang, Y., He, P., Li, H., Li, G., Liu, J., Jiao, F., Zhang, J., Huo, Y., Shi, X., Su, R., Ye, N., Liu, D., Yu, R., Wang, Z., Zhou, M., & Jiao, N. (2019). Ulva prolifera green-tide outbreaks and their environmental impact in the Yellow Sea, China. National Science Review, 6(4), 825–838. https://doi.org/10.1093/NSR/NWZ026